|
8/28/2023 0 Comments Define atomic number 
For example, Z 20 is always calcium, and calcium always has Z 20. However, once Z is known, the element is known hence, Z and X are redundant. X is the symbol for the element, such as Ca for calcium. You will need to refer to a periodic table for proton values. The number of protons in a nucleus is the atomic number Z, as defined in Medical Applications of Nuclear Physics. Atomic numbers are always whole numbers greater than zero, such as 1,2. of neutrons you can subtract the mass number (23) from the atomic number(11)). An atomic number of an element is the number of protons in all the atoms of that element. In this notation, the atomic number is not included. Every sodium atom has 11 protons, 12 neutrons and 11 electrons. For example, most noble gases have names ending with -on, while most. 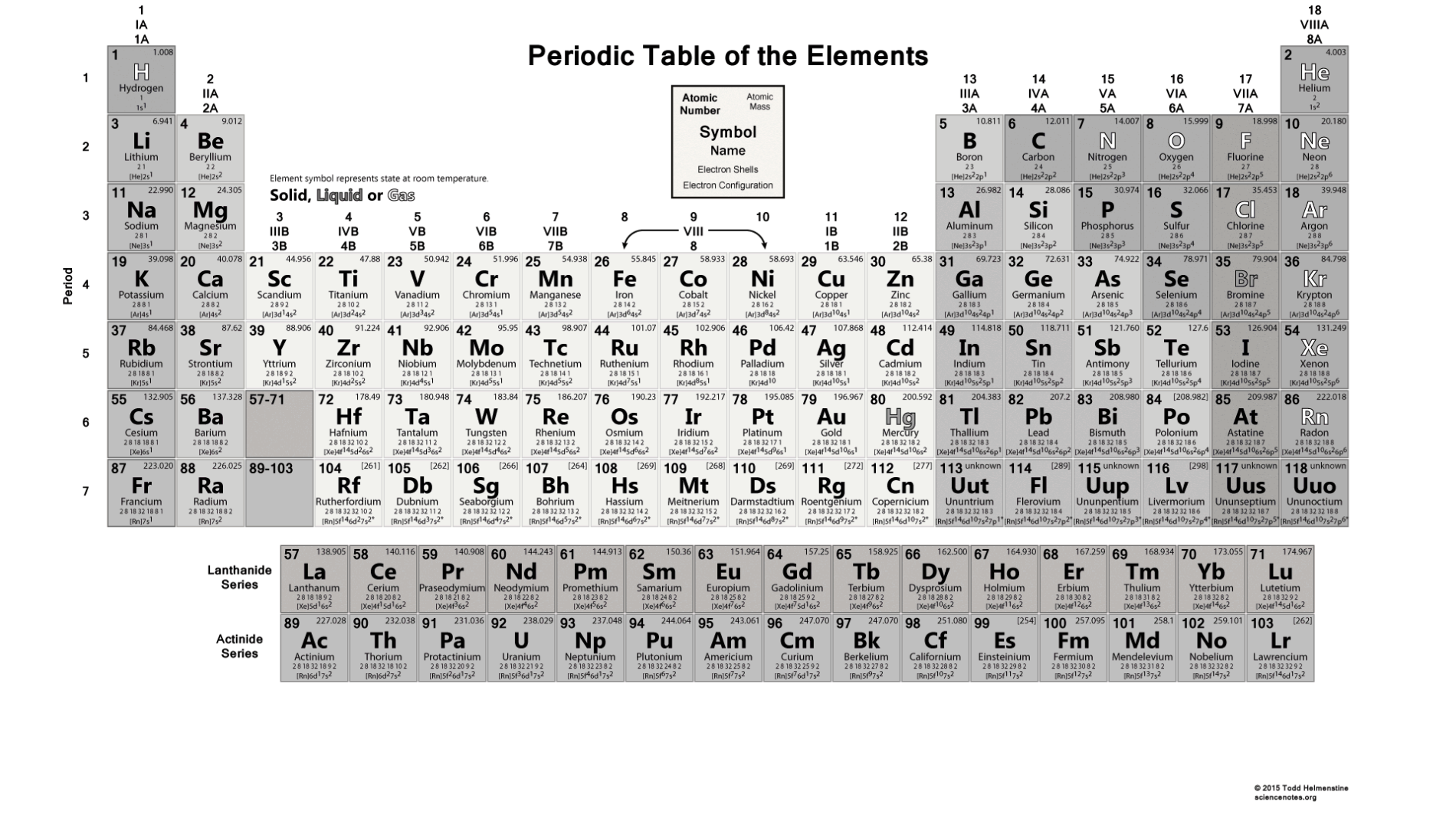
The names of some elements indicate their element group. If there is a second letter, it is lowercase. Each element has a symbol, which is one or two letters. Symbol-mass format for the above atom would be written as Cr-52. The atomic number or nuclear charge number (symbol Z) of a chemical element is the charge number of an atomic nucleus. The periodic table lists the elements in order of increasing atomic number. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. 100 Fm Fermium (257) 101 Md Mendelevium (258) 102 No Nobelium (259) 103 Lr Lawrencium (266) 104 Rf Rutherfordium (267). Since the iodine is added as a 1 anion, the number of electrons is 54. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 53 74). Both the atomic number and mass are written to the left of the chemical symbol. The atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. The meaning of ATOMIC NUMBER is an experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
